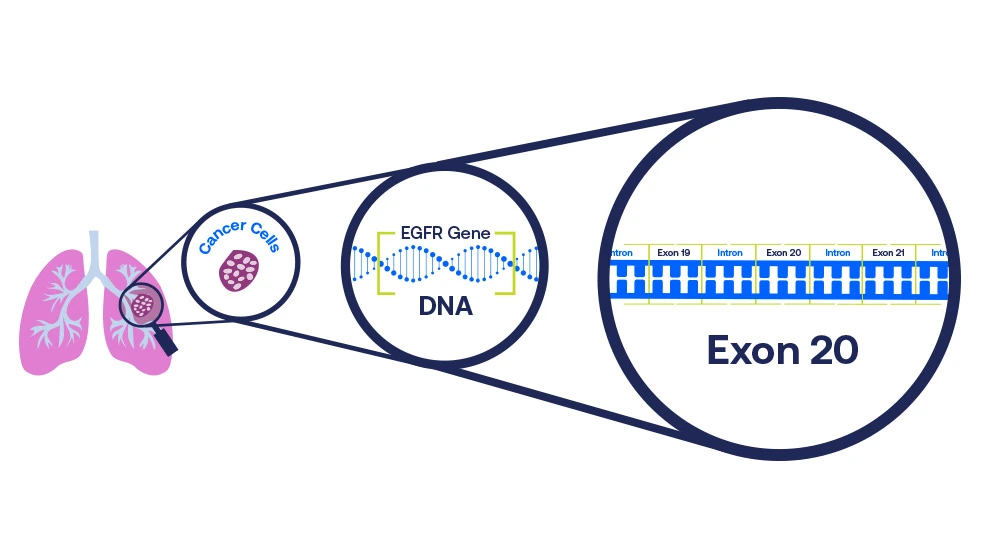
What is an Exon 20 insertion mutation?
EGFR exon 20 insertions are one type of lung cancer biomarker. They are a type of EGFR mutation that leads to uncontrolled cell growth and development of cancer.
Who is likely to have an EGFR exon 20 insertion mutation?
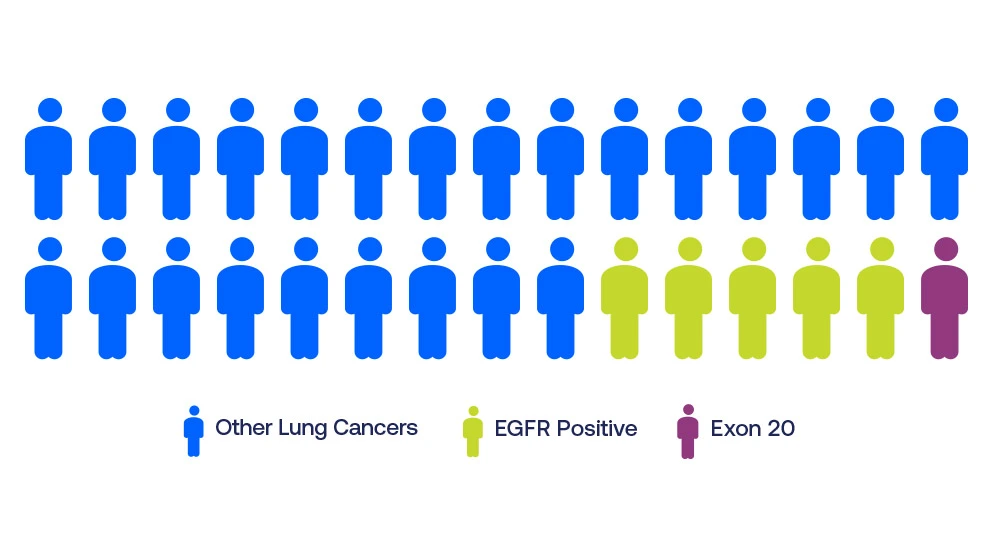
Patients who test positive for an EGFR mutation may have an exon 20 insertion mutation. The true frequency of EGFR exon 20 insertion mutations in non-small cell lung cancer (NSCLC) is unknown, but likely ranges from 1-10%, with 3-4% being most commonly reported. The mutation is more often found in people who never smoked and in Asian populations.
How do you know if you have an EGFR exon 20 insertion mutation?
EGFR exon 20 mutations can sometimes be difficult to detect with traditional biomarker testing techniques known as polymerase chain reaction-based markers (PCR). Newer and more comprehensive biomarker tests known as next-generation sequencing (NGS) can often have a higher chance of detecting these mutations.
Learn more about the different types of biomarker tests.
What are the treatment options for EGFR exon 20 insertion mutation patients?
Exon 20 insertion mutations are resistant to standard EGFR tyrosine kinase inhibitors (TKIs), which are drugs that are used to block cancer growth. If you are diagnosed with the EGFR exon 20 mutation, your first-line treatment option will likely include chemotherapy with amivantamab (Rybrevant), a type of targeted drug called a monoclonal antibody. Sunvozertinib (Zegfrovy) is also an FDA-approved treatment for EGFR exon 20 insertion NSCLC post-platinum chemotherapy (approved in July 2025).
If your cancer grows after treatment, you should get another biopsy to see if a different biomarker has developed. If a new biomarker has been identified, your doctor will recommend a targeted therapy to treat that biomarker.
If no new biomark is found, your doctor may recommend one of the following options:
- Chemotherapy alone or with amivantamab (if you haven’t already received it)
- Chemotherapy with another monoclonal antibody called bevacizumab (Avastin)
- Immunotherapy
All lung cancer patients should ask their doctors if they are eligible to participate in a clinical trial.
What Should You Do Next?
If you are diagnosed with NSCLC, but have not received biomarker testing, ask your doctor about comprehensive biomarker testing.
Work with your doctor to discuss your goals and options each time you have to make a treatment decision. The three big questions to ask are:
- What is the goal of this treatment?
- What are the potential side effects?
- What other options do I have?
Research is happening at a rapid pace, and your doctor should be up to date on the recommendations for your specific type of lung cancer. If you don’t feel comfortable with the answers you are receiving, do not hesitate to seek out a second opinion.
Page last updated: February 24, 2026